The fire inside

In the first of a two-part series, JAKE VENTER explains the basics of combustion.
Generally speaking, engines can be divided into two groups: external combustion engines and internal combustion engines. Steam engines are classified as external combustion engines because they rely on a fire outside the engine to supply the high pressure fluid that’s needed to develop a worthwhile torque. These engines generally generate such a high average pressure on the pistons that they develop maximum torque at start-up. In fact, steam locomotives don’t need a gearbox, and many of them have only two cylinders!
Petrol and diesel engines are known as internal combustion engines because the fire is inside. This results in a fairly low average pressure on the pistons, so that vehicles with internal combustion engines always need a gearbox to multiply the engine’s torque.
We’ll take a look at the combustion process in these engines, but are again faced with a choice. While petrol engine combustion takes place in a fuel/air mixture as a result of a spark, diesel engine combustion takes place in air because fuel is injected into a hot environment.
PETROL ENGINE COMBUSTION
This article will focus on petrol engine combustion, leaving diesel engine combustion for the next instalment. Combustion is a chemical reaction that takes place between fuel and oxygen. In this case, the fuel is a mixture of hydrocarbon compounds that have been formulated to burn controllably 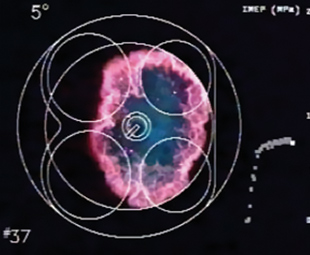 but not explode when a spark occurs. The octane number of petrol is a measure of this good behaviour, but we won’t explore this any further now. Perhaps it would make a good topic for another article…
but not explode when a spark occurs. The octane number of petrol is a measure of this good behaviour, but we won’t explore this any further now. Perhaps it would make a good topic for another article…
Back to combustion. During the one or two milliseconds (ms) that it takes for combustion to be completed, a number of processes take place:
1. The hydrogen in fuel combines with oxygen in the air to form water at the rate of approximately one litre of water for every litre of fuel burnt. When an engine is cold one can usually see water dripping out of the exhaust pipe, but later the water exits as invisible steam.
2. If the fuel/air mixture is perfect, the carbon in the fuel combines with oxygen in the air to form carbon dioxide. This clear, odourless gas is also what humans and animals exhale.
3. However, because the mixture never is perfect the carbon forms carbon monoxide: an odourless but harmful gas that puts you into a permanent sleep. The amount of carbon monoxide produced is closely connected to the amount of excess fuel (rich mixture) being supplied.
4. Small amounts of nitrogen- and sulphur-oxides are also formed from the nitrogen in the air and a sulphur impurity in the fuel. In addition, there are usually traces of unburnt hydrocarbons present because of excess fuel that did not combust. These components, as well as the carbon monoxide, are to a large extent rendered harmless by the catalytic converter.
Now that we know what is being produced inside the combustion chamber, it is time to take a look at what’s happening during one combustion event in an engine that’s doing a constant 3 000 r/min. Let’s assume that we can see through a toughened window into the space above the piston.
At 20° before top dead centre (TDC), when the piston is 1.11 ms away from the top, the ignition system delivers 30 000 volts of electrical energy to the spark plug, which fires a spark across its gap. This initiates the first phase of combustion – ignition and flame development. At about 14° before TDC (0.35 ms after the spark occurred) the young flame becomes visible as a string shape between the electrodes and starts to spread. This phase consumes about 5% of the fuel/air mixture, but there is not yet any rise in pressure in the space above the piston.
The next phase – flame propagation – starts about 0.35 ms later, when the piston is 0.41 ms away from TDC and the heat from the greatly enlarged flame starts to produce a very sharp rise in pressure. In a closed container of still air this flame would have travelled in a ball-shape at about 30 km/h to the end of the combustion chamber. However, the violent motion of the air above the piston – caused by the combustion chamber shape as well as the piston’s motion – has not only distorted the shape but has also speeded-up the flame speed to about 300 km/h. This is just as well, because the piston has by now passed TDC and is on its way down to start the power stroke. The maximum temperature of over 3 300 °C and maximum pressure of approximately six times its value at the beginning of the compression stroke both occur at about 10° (0.555 ms) after TDC. The flame termination phase occupies another 10° of crankshaft motion before the last flame dies against the walls of the combustion chamber.
Although this entire process has taken only a few ms, the pressure kick from each combustion stroke is enough to keep the engine and its load going for as long as it gets a mixture and a spark.
Published by
Focus on Transport
focusmagsa




